 A concentrated structure of positive ions shares loosely spread electrons in metallic bonding. The binding of atoms in a metallic cluster is totally different from that of ionic binding formed by linking opposite polarity ions or covalent binding formed by sharing electrons. Here again, the metallic cluster is the differentiating element. In the scenario of covalent bonds also, the atomic radii concept remains the same, except that the chemical compound is formed by sharing electrons. The distance between the nucleus of a positive ion and the nucleus of a negative ion is considered as twice the radius of an ion. This bond exists not in an elemental state but in a chemical compound state. The ionic bond is a type of electrovalent bond, where two oppositely charged ions attract each other to form a link. See how these two non-bonded atoms are themselves bonded into a compound molecule in the figure below. This concept is defined as the distance between the nucleus of two neighbouring atoms which are in non-bonded conditions. Let us see each of these concepts in detail below. The approximation of a circle or a sphere is one way. As said earlier, it is difficult to exactly predict the shape and construct of an atom, due to limited facts available. There are many types of atomic radius concepts prevalent. Thus the separation from the center of the core to the exterior shell of an atom is called an atomic radius. 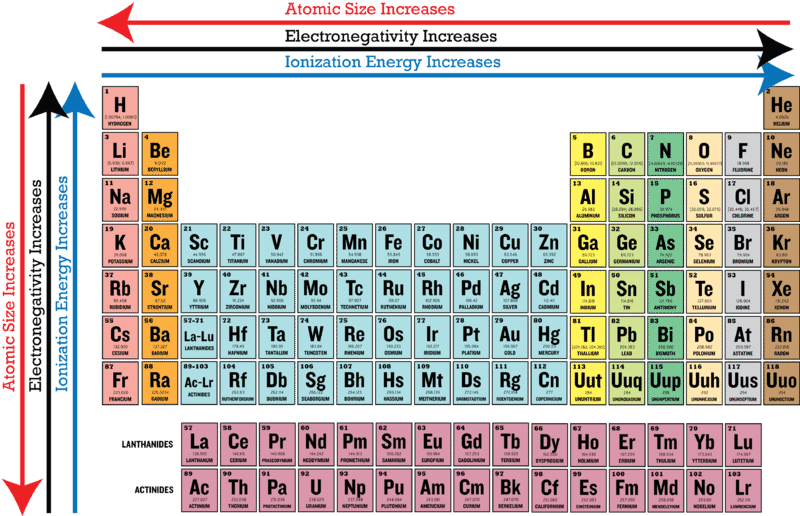
An atom contains different shells in circle form, one over the other, and hence the radius of an atom is measured with the isolated electron in the outermost circle as portrayed below. The diameter is a line between two points on the circle and passing through the center and radius it's half. How is the radius of a circle defined and how different is the atomic radius defined? A sphere is a circle projected in the third dimension. Secondly, atomic radius is characterized by approximating the appearance of an atom as a sphere. Please refer to the figure below.įirstly, radius as a term is related to a geometrical figure circle. From top to down, both atomic number and size increase. But the atomic size in the periodic table decrease as you go from left to right. The atomic number from left to right of a periodic table increase progressively. Atomic radius is defined as the measurement of the distance from the nucleus of an atom to the outermost electron available in the shell in isolated conditions. The quantity of protons in the core of molecule is the atomic number. It contains elements arranged with the smaller atomic number to the highest atomic number in different groups. 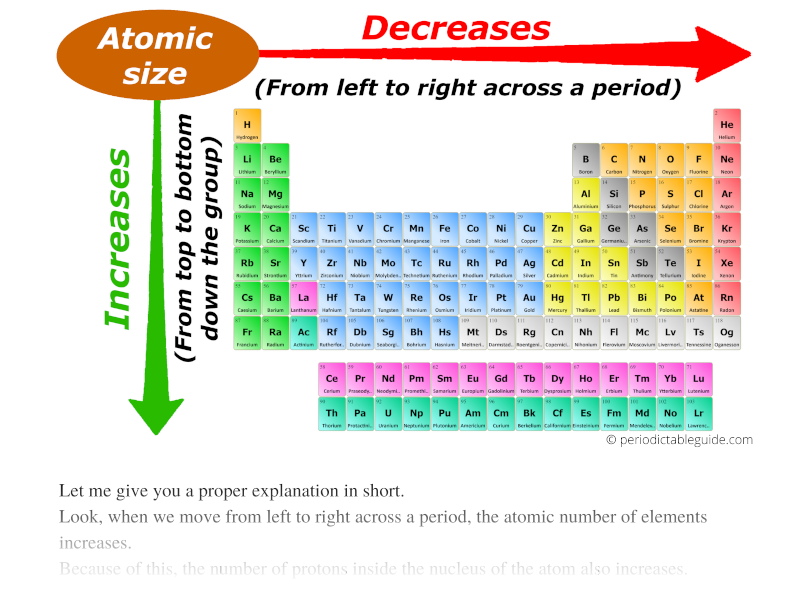
The periodic table is an organized table for reference. The periodic table patterns, different types of radii and so on, are discussed below. This article covers all aspects of atomic radii of chemical elements. The smaller the radii, they will be more reactive than the larger ones. Knowing the atomic radius helps us to predict the reactivity levels of atoms. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
